GLP-1
GLP-1: The Bridge between
Glucose, Satiety and Body Weight
In the current landscape of metabolic health, GLP-1 occupies a central position. Its growing prominence within the scientific community has been marked by the development of drugs targeting its receptor. However, beyond its therapeutic application, a deeper question emerges: What is the role of GLP-1 as a regulator of metabolism?
Far from being merely a “weight-loss hormone,” glucagon-like peptide-1 (GLP-1) is a metabolic integrator that connects glucose regulation, appetite control and energy balance (1,2). Understanding this axis allows for a more systemic view of metabolism.
A Hormone with a Coordinated Function
GLP-1 is an incretin hormone produced by enteroendocrine L cells, primarily located in the ileum and colon. After secretion, it circulates in two main active forms GLP-1 (7–36) and GLP-1 (7–37), with a short half-life due to rapid degradation by the enzyme DPP-IV (1).
Its biological action depends on binding to the GLP-1 receptor (GLP-1R), which is expressed in multiple tissues. Activation of this receptor triggers intracellular cAMP-dependent signaling, that (1,3):
- Stimulates glucose-dependent insulin secretion
- Inhibits glucagon release
- Preserves functional pancreatic β-cell mass
- Improves postprandial glycemic homeostasis
This mechanism positions GLP-1 as a central modulator of metabolic control.
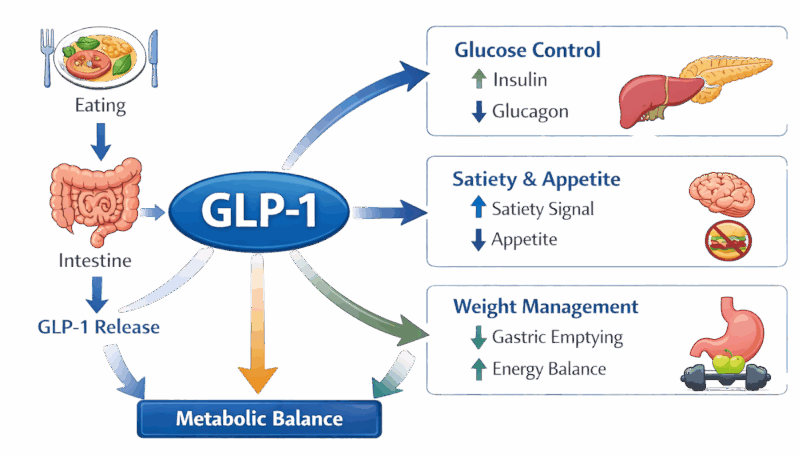
Conceptual scheme based on (Wang et al., 2021; Domínguez-Avila et al., 2017; Derosa et al., 2025).
The Starting Point:
Glucose Regulation
Amid the widespread media attention surrounding GLP-1 analogues such as semaglutide (Ozempic) and liraglutide (Saxenda), public discourse has largely centered on their effects on body weight reduction. However, this emphasis has somewhat eclipsed the primary physiological role of GLP-1: the regulation of postprandial glucose, namely the control of the rise in blood glucose levels following food intake.
A more stable postprandial glycemic response reduces both metabolic variability and compensatory hyperinsulinemia, two factors closely linked to the progressive development of insulin resistance and type 2 diabetes. In this context, the critical issue is not solely the magnitude of glucose elevation, but the efficiency with which the body regulates and adapts to that increase (2,4,5).
Type 2 diabetes does not emerge abruptly; rather, it develops gradually as peripheral tissues become less responsive to insulin and pancreatic β-cells undergo progressive functional decline. When these processes converge, the maintenance of glycemic homeostasis becomes compromised.
Within this framework, the incretin system, particularly the GLP-1/GLP-1R axis, assumes significant physiological importance (1,3,6). GLP-1 enhances glucose-dependent insulin secretion, meaning its action is amplified when glucose concentrations rise after a meal, while concurrently suppressing glucagon release. This coordinated response enables a more physiologically appropriate metabolic adjustment, contributing to the preservation of β-cell function and improved overall metabolic regulation (1,6).
Accordingly, strategies aimed at optimizing postprandial glycemic control extend beyond simple weight reduction. They seek to enhance the quality and efficiency of the metabolic response to each meal, an approach with broader implications for the prevention and long-term management of metabolic disorders (2,7,8).
GLP-1 and Satiety:
The Gut-Brain Connection
Beyond its role in glycemic control, GLP-1 directly participates in appetite regulation. This hormone slows gastric emptying, prolonging the sensation of fullness after meals and acts on hypothalamic neurons expressing GLP-1R, modulating satiety signaling and reducing food intake (1).
This effect is part of a feedback system that adjusts energy balance according to nutritional status. During fasting, ghrelin levels increase and stimulate appetite; after food intake, GLP-1 secretion rises and helps reduce this orexigenic signal. The reciprocal interaction between these hormones reflects coordinated regulation of energy balance, demonstrating that GLP-1 responds not only to glucose but to the organism’s overall metabolic state (1).
Microbiota and GLP-1
Fermentation of dietary fiber by the gut microbiota generates short-chain fatty acids (SCFAs), which can modulate GLP-1 secretion (6). This relationship reinforces the role of the intestine as a key endocrine organ in metabolic regulation and positions the diet–microbiota–incretin interaction as a relevant axis in cardiometabolic prevention (8,9).
Regulation of GLP-1 Secretion: A Multifactorial System
GLP-1 secretion responds to an integrated system of neuronal and nutritional signals.
Following food intake, an early phase occurs mediated by vagal activation and neuroendocrine mechanisms, followed by a second phase dependent on the direct contact of nutrients with the intestine (1). In this latter phase, glucose, fatty acids, microbiota-derived SCFAs and peptides or amino acids stimulate intestinal L cells through specific receptors and transporters (1,7).
This model confirms that GLP-1 functions as a metabolic sensor of nutrient intake.
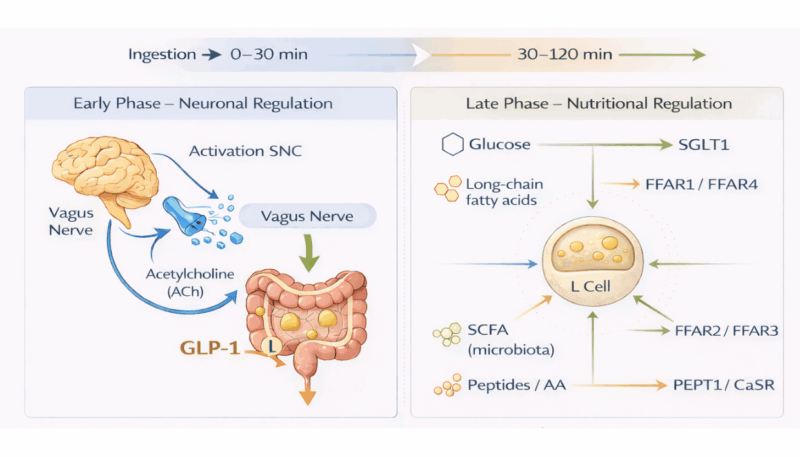
Adapted from (Wang et al., 2021; Domínguez-Avila et al., 2017; Iatcu et al., 2024).
Although its primary role is glycemic control, GLP-1 also exerts effects in other tissues with potential implications for hepatic function, cardiovascular regulation and bone metabolism (1). Overall, the GLP-1/GLP-1R axis can be understood as an integrative node connecting the intestine, pancreas and central nervous system in the regulation of metabolic balance.
Are you looking for the right ingredients?
Subscribe to our newsletter…
…or find our ingredients in the following product portfolio!
References
- Wang Y, Alkhalidy H, Liu D. The emerging role of polyphenols in the management of type 2 diabetes. Molecules. 2021;26(3):703.
- Derosa G, et al. Nutraceuticals and supplements in the management of prediabetes and diabetes: Mechanistic and clinical evidence. Nutrients. 2025;17(1):14.
- Domínguez Avila JA, et al. The antidiabetic mechanisms of polyphenols related to increased GLP-1 and insulin signaling. Molecules. 2017;22(6):903.
- Lown M, Fuller R, Lightowler H, Fraser A, Gallagher A. Mulberry-extract improves glucose tolerance and decreases insulin concentrations in normoglycaemic adults: A randomized double-blind placebo-controlled study. PLoS ONE. 2017;12(2):e0172239.
- Thondre PS, Lightowler H, Ahlstrom L. Mulberry leaf extract improves glycaemic response to sucrose in healthy subjects: A randomized, double-blind, placebo-controlled study. Nutrition & Metabolism. 2021;18:42.
- Abiola JO, et al. Potential role of phytochemicals as GLP-1 receptor agonists in diabetes. Pharmaceuticals. 2024;17(6):736.
- Iatcu OC, et al. Harnessing prebiotics to improve type 2 diabetes outcomes: Mechanisms and clinical relevance. Nutrients. 2024;16(20):3447.
- Venkatakrishnan K, et al. Functional foods and nutraceuticals in obesity and metabolic disorders. Food & Function. 2019;10:1240–1263.
- Cevallos-Fernández E, et al. Fermented plant-based foods and postbiotics for glycemic control: Mechanistic insights and clinical implications. Frontiers in Nutrition. 2026.